Ralston SH, Fraser J. Diagnosis and management of osteoporosis. Practitioner 2015;259 (1788):15-19
Diagnosis and management of osteoporosis
22 Dec 2015
AUTHORS
Professor Stuart H Ralston MD FRCP, Professor of Rheumatology, Centre for Genomic and Experimental Medicine, University of Edinburgh, Western General Hospital, Edinburgh, UK
Dr Jamie Fraser BSc MB ChB MRCGP, GP, Inverness, UK
Article
Osteoporosis is a common condition characterised by low bone mineral density(BMD) and an increased risk of fragility fractures.
It affects up to 30% of women and 12% of men at some point in their lives. The threshold for osteoporosis is defined operationally as BMD at the spine or hip 2.5 SD or more below the population average in young healthy individuals (T-score <-2.5), where BMD has been assessed by dual energy X-ray absorptiometry (DEXA).1
A large number of factors have been identified that increase the risk of osteoporosis and/or fragility fractures, see table 1. Two of the most important risk factors are increasing age and female gender, although other common and potentially modifiable risk factors include long-term corticosteroid therapy, chronic inflammatory disease, malabsorption and untreated premature menopause.
Clinical presentation
Osteoporosis is a clinically silent disease and may not present until it is quite advanced since low levels of BMD do not cause symptoms. A common presentation is with a low trauma fracture, often of the spine, wrist, or hip although almost any bone can fracture as the result of osteoporosis.
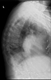
Peripheral fractures are easily diagnosed, but vertebral fractures (see figure 1) are easily missed. Sometimes they present with back pain after lifting a heavy object but often the presentation may be more insidious with chronic back pain, height loss and kyphosis. Height loss of more than 5 cm should raise the suspicion of vertebral fractures.
While fractures are the hallmark of osteoporosis, the majority of patients with fragility fractures do not have osteoporosis, at least as defined by low BMD on DEXA examination.2
Clinical assessment
History and examination
The patient should be asked about:
• Family history of osteoporosis (particularly parental hip fracture)
• General medical history, focusing on possible secondary causes of osteoporosis
• Smoking
• Alcohol intake
• Previous fracture
• Age at menopause
• Height loss
Clinical examination should include height and weight to assess body mass index and check for height loss. Other clinical signs may include kyphosis in patients with multiple vertebral fractures and local tenderness on percussion over the site of a recent vertebral fracture.
Fracture risk assessment
The diagnosis of osteoporosis can be confirmed by DEXA but this should only be performed in patients who have an increased risk of fracture on the basis of clinical risk factors.3,4 Fracture risk assessment is seldom indicated in patients under the age of 50 unless there are very strong risk factors, see figure 2.
Fracture risk can be estimated in several ways but the two most commonly used tools are QFracture (www.qfracture.org/) and FRAX (www.shef.ac.uk/FRAX/).
The QFracture tool has been extensively validated in the UK and takes account of information from a range of clinical risk factors as well as ethnicity. Fracture risk can be calculated in patients aged 30-99 years over various time scales between one and ten years. Many GP computer systems offer the option of QFracture as an add-on allowing automatic calculations from information already held in the electronic patient record.
The FRAX calculator uses a smaller number of risk factors and operates over an age range of 40-90 years. The FRAX tool is less well validated in the UK population and the algorithm underlying the calculation is unpublished. The calculator takes life expectancy into account and because of this FRAX may underestimate actual risk over shorter timeframes in the elderly.5 An advantage of FRAX is that it can calculate fracture risk taking account of hip BMD, as opposed to QFracture which does not offer individual assessment using bone density.
A limitation of both tools is that they underestimate the risk of vertebral fractures.
Bone densitometry
Bone densitometry by DEXA is helpful in confirming the diagnosis and for assessing who will benefit from treatment. According to recent SIGN guidance4 evaluation by DEXA should be considered if the ten-year risk of major osteoporotic fracture is more than 10%. The rationale for this recommendation stems from clinical trial data which has shown evidence of benefit for treatment in patients with osteoporosis on DEXA who have a fracture risk > 10%.6,7
The NICE fracture risk assessment guidance3 did not suggest a threshold at which DEXA should be considered.
If the BMD T-score values by DEXA at the lumbar spine, femoral neck or total hip are at or below -2.5 then the diagnosis of osteoporosis is confirmed.
Vertebral fractures are generally taken as diagnostic of osteoporosis, even if spine BMD values are not in the osteoporotic range. This is particularly relevant in elderly patients where co-existing osteoarthritis and aortic calcification are common resulting in falsely elevated spine BMD values.
While DEXA is not strictly necessary in patients with vertebral fracture it is helpful in providing a baseline upon which to assess response. This is useful in patients who suffer a fracture during treatment (see below).
Other investigations
Other investigations are indicated to determine if there is an underlying cause for the osteoporosis and to determine if there are any contraindications to specific treatments. A typical screen includes tests for coeliac disease (TTG antibody), thyrotoxicosis (TFT), myeloma and monoclonal gammopathy of uncertain significance (serum protein electrophoresis and urinary Bence-Jones protein); renal impairment (urea and electrolytes;
liver disease and alcohol excess (liver function tests), osteomalacia and hyperparathyroidism (serum calcium phosphate) and male hypogonadism (serum testosterone and gonadotrophins).
In our experience the most common secondary causes of osteoporosis are occult coeliac disease, monoclonal gammopathy of uncertain significance and hypogonadism.
Management
The management of osteoporosis consists of identifying and treating any underlying causes, correcting lifestyle factors that might contribute to the osteoporosis or risk of fractures and instituting drug treatment where appropriate, see table 2. Although correction of secondary causes of osteoporosis is generally beneficial, expert advice should be sought before initiating testosterone replacement in male patients with hypogonadism because of the increased risk of cardiovascular disease.8
Who should be treated?
The best evidence is in postmenopausal osteoporosis where various treatments have been shown to reduce the risk of further fractures in patients with osteoporosis on DEXA and/or those with pre-existing low trauma vertebral fractures.
Some treatments have been found to prevent fractures even in patients who have not been shown to have osteoporosis on DEXA. They include HRT in postmenopausal women;9 intravenous zoledronic acid in elderly patients following a hip fracture10 and calcium and vitamin D supplements in nursing home residents.11
Contrary to previous NICE guidance however, there is no clinical evidence to support the use of anti-osteoporosis medications to prevent further fracture in elderly patients with a history of fragility fracture. Although some of these patients do have osteoporosis, the majority do not and are unlikely to benefit from treatment.12 Indeed, previous studies have shown that alendronic acid does not prevent fractures in osteopenic women13 and that risedronate does not prevent hip fractures in elderly patients with clinical risk factors alone.14
Choice of treatment
Oral bisphosphonates are the first-line treatment. The most widely used agent is oral alendronic acid, 70 mg once a week with calcium and vitamin D supplements. Oral risedronate 35 mg once a week has similar anti-fracture efficacy.
The most common adverse effect with oral bisphosphonates is upper gastrointestinal upset, although many patients who develop GI upset with alendronate can take risedronate perfectly well and vice versa.
Hiatus hernia, gastro-oesophageal reflux and Barrett’s oesophagus are not contraindications to oral bisphosphonates.
If oral bisphosphonates are contraindicated or not tolerated then parenteral therapy should be considered. The first-line treatment is intravenous zoledronic acid 5 mg once a year, although denosumab 60 mg every six months subcutaneously is equally effective.
Parenteral therapy is usually initiated in secondary care. It is common for patients to experience a transient flu-like illness for 2-3 days after the first dose of zoledronic acid, but this does not usually recur with subsequent infusions. There is a risk of hypocalcaemia with both parenteral preparations but that can be minimised by ensuring that the patient is given calcium and vitamin D supplements prior to and during therapy.
Unlike bisphosphonates, denosumab can be used in patients with renal impairment but great care must be exercised in this patient group because of the high risk of hypocalcaemia which may be serious or even fatal. These patients are best treated in secondary care.
Teriparatide (the 1-34 fragment of parathyroid hormone; TPTD) is highly effective in the treatment of vertebral osteoporosis and has been shown to be superior to oral bisphosphonates in preventing vertebral fractures, both in randomised trials and observational studies.15
Therapy with TPTD is usually initiated in secondary care either as primary treatment for severe osteoporosis or as rescue therapy in patients who have failed to respond to other agents. Following a two-year course patients are switched to antiresorptive therapy to maintain the increase in BMD.
Duration of therapy
It is unclear how long patients should remain on treatment. This is because drugs such as bisphosphonates and denosumab suppress the normal process of bone renewal and repair.
For most drugs there is good evidence of safety and efficacy for up to five years. After that point the need for ongoing therapy should be reviewed.16
If the patient still has osteoporosis or has suffered fractures there may be benefit in continuing therapy for another five years in patients at high risk of vertebral fracture.17
For zoledronic acid most centres give a course of three years and a spell off therapy for three years based on research which showed no difference in clinical fracture rates in patients treated for three years compared with six.18
Long-term adverse events related to oversuppression of bone turnover include atypical subtrochanteric fractures and osteonecrosis of the jaw, although these are very rare and in the vast majority of patients the benefits of therapy outweigh the risk. There is no evidence that ‘drug holidays’ reduce the risk of the complications related to oversuppression of bone turnover.
Patients who fracture while on treatment
None of the treatments currently available are completely effective at preventing fractures. The results are best for vertebral fractures, relative risk (RR) reduction 50-75%; quite good for hip fractures (RR reduction 40%) but modest for non-vertebral, non-hip fractures (RR reduction 20-25%).
Some patients may suffer fractures while on anti-osteoporosis therapy.
If this occurs it is important to check compliance (and with oral bisphosphonates to make sure the treatment is being taken when the patient has fasted). If the medication is being taken correctly treatment should be continued and the patient informed that fractures can still occur even though the treatment is working.
If two or more fractures occur during therapy, then a repeat BMD measurement should be requested provided at least two years has elapsed since the last scan. Measurements at shorter intervals are seldom informative because of limitations in the precision of DEXA. If the repeat DEXA shows significant bone loss (more than 4%) then an alternative treatment should be considered. Most often this will involve changing from an oral to a parenteral medication.
Special groups
The management of male osteoporosis follows the same general strategy as for postmenopausal osteoporosis. The evidence base for fracture prevention in men with osteoporosis is poor, since most drugs have been licensed for the treatment of male osteoporosis on
the basis that they increase BMD, rather than prevent fractures. The best evidence for anti-fracture efficacy in men is with zoledronic acid for the prevention of vertebral fractures.19
Similar comments apply to glucocorticoid-induced osteoporosis (GIOP), although here there is evidence that bisphosphonates and TPTD are effective at preventing vertebral fractures, when combined with calcium and vitamin D supplements. The entry criteria for clinical trials in GIOP studies generally focused on patents who were on 7.5 mg prednisolone or greater for more than three months and this is often the trigger for initiating bone protective therapy in routine practice.
There is evidence that fractures occur in GIOP at higher levels of BMD than in postmenopausal osteoporosis so therapy should be considered in patients with a BMD T-score of <-1.5.
Although it is useful to have a DEXA scan before starting treatment to provide a baseline value to assess response, this investigation is not absolutely necessary to initiate bone protective therapy especially in those aged above 65 since the vast majority of these patients will have a T-score of -1.5 or below. In younger individuals where BMD is likely to be higher DEXA is useful in determining if bone protective treatment is needed immediately or if it could be delayed until the T score falls below -1.5.
REFERENCES
1 Kanis JA, Melton LJ III, Christiansen C et al. The diagnosis of osteoporosis. J Bone Miner Res 1994;9(8):1137-41
2 Siris ES, Miller PD, Barrett-Connor E et al. Identification and fracture outcomes of undiagnosed low bone mineral density in postmenopausal women: results from the National Osteoporosis Risk Assessment. JAMA 2001;286(22):2815-22
3 National Institute for Health and Care Excellence. CG146. Osteoporosis: assessing the risk of fragility fracture. NICE. London. 2012 www.nice.org.uk
4 Scottish Intercollegiate Guidelines Network. SIGN 142. Management of osteoporosis and the prevention of fragility fractures. SIGN. Edinburgh. 2015 www.sign.ac.uk
5 Bolland MJ, Jackson R, Gamble GD, Grey A. Discrepancies in predicted fracture risk in elderly people. BMJ 2013;346:e8669
6 Donaldson MG, Palermo L, Ensrud KE et al. Effect of alendronate for reducing fracture by FRAX score and femoral neck bone mineral density: the Fracture Intervention Trial. J Bone Miner Res 2012;27(8):1804-10
7 Cummings SR, San MJ, McClung MR et al. Denosumab for prevention of fractures in postmenopausal women with osteoporosis. N Engl J Med 2009;361(8):756-65
8 Xu L, Freeman G, Cowling BJ, Schooling CM. Testosterone therapy and cardiovascular events among men: a systematic review and meta-analysis of placebo-controlled randomized trials. BMC Med 2013;11:108
9 Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the Women's Health Initiative randomized controlled trial. JAMA 2002;288(3):321-333
10 Lyles KW, Colon-Emeric CS, Magaziner JS et al. Zoledronic acid and clinical fractures and mortality after hip fracture. N Engl J Med 2007;357(18):1799-1809
11 Chapuy MC, Arlot ME, Duboeuf F et al. Vitamin D3 and calcium to prevent hip fractures in the elderly women. N Engl J Med 1992;327(23):1637-42
12 Ralston SH, de'Lara G, Farquhar DJ. NICE on osteoporosis. Women over 75 with fragility fractures should have DEXA. Br Med J 2009;338:b2340
13 Black DM, Thompson DE, Bauer DC et al. Fracture risk reduction with alendronate in women with osteoporosis: the Fracture Intervention Trial. FIT Research Group. J Clin Endocrinol Metab 2000;85(11):4118-24
14 McClung MR, Guesens P, Miller PD et al. Effect of risedronate on the risk of hip fracture in elderly women. N Engl J Med 2001;344(5):333-40
15 Oswald AJ, Berg J, Milne G, Ralston SH. Teriparatide treatment of severe osteoporosis reduces the risk of vertebral fractures compared with standard care in routine clinical practice. Calcif Tissue Int 2014;94:176-82
16 Drug Safety Update. MHRA. 2011
17 Ensrud KE, Barrett-Connor EL, Schwartz A et al. Randomized trial of effect of alendronate continuation versus discontinuation in women with low BMD: results from the Fracture Intervention Trial long-term extension. J Bone Miner Res 2004;19(8):1259-69
18 Black DM, Reid IR, Boonen S et al. The effect of 3 versus 6 years of zoledronic acid treatment of osteoporosis: a randomized extension to the HORIZON-Pivotal Fracture Trial (PFT). J Bone Miner Res 2012;27(2):243-54
19 Boonen S, Reginster JY, Kaufman JM et al. Fracture risk and zoledronic acid therapy in men with osteoporosis. N Engl J Med 2012;367(18):1714-23
ARTICLE IN PDF
PRESET SEARCHES
NICE Evidence
PubMed
Osteoporosis treatment primary care
Immediate CPD prompts
These handy templates* prompt immediate drafting of personal learning, reflection and planning. Save your record straight into your personal development folder on your device or Cloud storage system.
*Adobe reader needed for iPad
EXTERNAL WEBLINKS
LETTERS TO THE EDITOR
If you would like to comment on this article or have a question for the authors, write to: [email protected]

 = Registered users
= Registered users = Paid-up subscribers
= Paid-up subscribers