Hopper AD, Campbell J A. Early diagnosis of oesophageal cancer improves outcomes. Practitioner 2016; 260(1791):23-28
Early diagnosis of oesophageal cancer improves outcomes
21 Mar 2016
AUTHORS
Dr Andrew D Hopper MD FRCP, Gastroenterology Consultant
Dr Jennifer A Campbell MRCP, Clinical Fellow in Gastroenterology, Department of Gastroenterology, Royal Hallamshire Hospital, Sheffield, UK
Article
More than 7,000 cases of oesophageal cancer were diagnosed in 2013 making it the twelfth most common cancer in the UK. However, its poor outcome makes it the sixth most common cause of cancer deaths.1
There are two main types, oesophageal squamous cell carcinoma (OSCC) and oesophageal adenocarcinoma (OAC). Although their pathogenesis differs they present in the same manner. Both carry a very poor five-year survival of 16% when compared with more common cancers such as colorectal (59%), prostate (84%) and breast cancer (88%).2 Worryingly, the UK has the highest incidence of OAC in Europe and has seen a 38% increase in cases in the past three years.3,4 OAC is now the more common form of oesophageal cancer seen in the UK, Australia and other Western developed countries. OSCC remains more common globally.
Like many cancers, improved survival requires early diagnosis. This review focuses on symptom recognition and risk factors to initiate early endoscopy referral and diagnosis that improves the outcome of this potentially curable cancer. Current evidence regarding available and appropriate treatment options are then reviewed.
Risk factors
UK cancer registration statistics show a 2:1 male to female ratio for oesophageal cancer. Peak incidence at presentation is in the 65-75 age group, with 95% of cases presenting in those over the age of 50.3 Smoking is a major risk factor for both types of oesophageal cancer and is linked to an estimated 66% of cases in the UK.3 OSCC is linked to alcohol, smoking and chewing betel quid.4
OAC is associated with the presence of GORD and its duration5 and obesity (especially increased waist circumference).6
Metaplastic change in the distal oesophagus from recurrent acid reflux damage, known as Barrett's oesophagus, is a precursor and risk factor for OAC. The risk of developing OAC with Barrett's oesophagus is currently 0.1-0.33% per year.7,8 Diagnosis of Barrett’s oesophagus generally triggers endoscopic surveillance to enable early diagnosis in the event of cancer developing which improves survival.9 GORD has been discussed in a previous review article in this journal.10
Suspicious symptoms
Oesophageal cancer commonly presents with dysphagia or odynophagia (pain with swallowing). This can be associated with weight loss and vomiting. Other important causes of dysphagia are listed in table 1, but referral for urgent endoscopy should still be considered in the presence of dysphagia regardless of previous history or medication. The significance of dysphagic symptoms was highlighted in a recent study based on symptom referral for rapid access endoscopy. Dysphagia, weight loss and age were strong positive predictors for cancer. In this study, 92% of patients with malignancy had either dysphagia, weight loss or were over the age of 55 with other alarm symptoms (see table 2).11 Although involuntary or unintentional weight loss has been defined as greater than 5% of body weight in over six months, in clinical practice objective markers are rarely available therefore any subjective history of weight loss in the absence of any known illness should be considered given its importance.12,13
Because of the elasticity of the oesophagus, advanced tumours can present without dysphagic symptoms. Anaemia (lesion bleeding), hoarse voice (early mediastinal invasion) or weight loss (metastatic spread) may manifest. At-risk or alarm symptoms for oesophago-gastric cancer have been identified in guidelines by NlCE,14 SIGN15 and the British surgical and gastroenterological societies.16
The NICE recommendations for endoscopy referral to assess for suspected oesophageal cancer in their recently updated guidelines are shown in table 2.
These guidelines differ slightly from other earlier guidelines regarding who to refer urgently or to consider for non urgent endoscopy. SIGN recommends early endoscopy for patients with dysphagia, recurrent vomiting, anorexia, weight loss or gastrointestinal (GI) blood loss regardless of age15 and the British surgical and gastroenterological societies recommend rapid access endoscopy
for all patients over 55 with recent onset dyspepsia regardless of a response to treatment or all patients with alarm symptoms irrespective of age.16
Given that the NICE guidance is the most up to date it should probably be used to guide the urgency of referral.
However, patients over 55 with dyspepsia should be fully reviewed to assess for the ‘full’ response to treatment. Given the current lack of other accurate diagnostic tests and the low complication rate of upper GI endoscopy non urgent referral for endoscopy is acceptable when any clinical suspicion is raised, persisting upper GI symptoms are unexplained or proton pump inhibitor (PPI) treatment is required long term (> six weeks).
Confirming diagnosis
Endoscopy
Upper GI endoscopy with biopsy is the recommended investigation for patients with dysphagia to confirm oesophageal cancer. Lesional biopsy with histological interpretation is required to identify cancer subtype and exclude other causes such as severe gastro-oesophageal reflux and ulceration, see table 1.
Repeat gastroscopy should be performed if histology is benign and endoscopic appearances were suspicious of cancer. In cases of severe reflux, gastroscopy with biopsies is repeated after six weeks of anti-acid treatment to ensure healing and exclude underlying cancer or Barrett’s oesophagus. Despite improved advances in endoscopic imaging, failure to diagnose gastric cancer at initial endoscopy is consistently around 10%. Therefore patients with unexplained symptoms may require a second gastroscopy.17,18 The principal factors associated with repeat gastroscopy include failing to suspect malignancy and misdiagnosing reflux oesophagitis or a peptic stricture at the first examination. Failure to take adequate biopsies can result in false-negative histology.
Over the counter availability of ranitidine and PPI medication means that patients may well be taking an anti-acid medication at presentation. Initial gastroscopy should follow a break in PPI therapy, although there is no evidence to suggest the best timing, two weeks is usually suggested.
PPIs may mask endoscopic findings and ‘heal’ malignant ulcers or alter their appearance. Barium studies can be performed if the patient is too unwell or keen to avoid gastroscopy.19 Sensitivity of barium is reasonable for detecting malignancy but does not allow histological sampling to differentiate between malignant and benign ulceration and diagnosis can be delayed.
Cancer staging
If a lesion suspicious of oesophageal cancer is seen at gastroscopy, the patient is warned and referral to a specialist upper GI surgery unit is made.
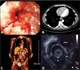
A thorough staging process is undertaken to allow patients to choose appropriate treatments and avoid patients with advanced or incurable disease undergoing unnecessary, significant surgery, see figure 1. Oesophageal cancer staging employs the Tumour, Nodal, Metastases (TNM) classification system, see table 3.20
Computed tomography (CT) of the chest abdomen and pelvis is performed initially to detect incurable disease and would usually be requested at the time of a suspicious endoscopy. It has a high (90%) sensitivity for detecting distant metastases > 1 cm and significant local invasion into adjacent mediastinal organs (85-100%).21-23
If distant metastases and local invasion are absent, a clinical assessment is required to discuss findings and assess suitability for curative treatment including surgery. Given the potential significant surgery, detailed below, and the likely age at presentation, a formal objective cardio-respiratory assessment is often required including exercise tolerance24 or complex cardiopulmonary exercise testing.2
Patients choosing curative treatment undergo complete TMN staging, see table 3. Endoscopic ultrasound (EUS) provides accurate assessment of the tumour size and local lymph node stage. This is important as tumours with early T stage (1-2) do not benefit from neoadjuvant chemotherapy before curative resection (see treatment section below).26
Positron emission tomography (PET) using F-18 fluorodeoxyglucose is also performed to detect distant lymph node or metastatic disease. Around 5% of patients with oesophageal cancer who are initially thought operable are precluded from oesophagectomy after CT and EUS staging.27
Staging laparoscopy is indicated where the tumour involves the lower oesophagus and upper stomach and CT shows potentially operable disease. Laparoscopy can detect peritoneal and metastatic disease under 5 mm in diameter, and enables peritoneal cytology and biopsies to be obtained from suspicious lesions. Staging laparoscopy changes treatment decisions for invasive surgery in up to 28% of patients with gastric cancer after CT.28 As with all cancers the stage of a cancer is closely related to prognosis emphasising the need to diagnose oesophageal cancer at an early stage, see table 4.29
The staging process highlights the complex pathway and investigations required before reaching a decision regarding suitability for curative surgery and treatment. If needless significant abdominal and thoracic surgery is performed in patients with advanced disease the recovery from surgery is likely to have a huge impact on the patient’s quality of life for their remaining life expectancy. Throughout the staging process it is imperative that patients are supported by cancer nurse specialists.
Management
Management options are discussed at a specialist upper GI multidisciplinary team (MDT) meeting involving experienced surgeons, radiologists, pathologists, oncologists and cancer nurse specialists. Investigation results are considered alongside the patient’s fitness for surgery and/or chemotherapy and the final decision made together with the patient after the clinician has explained the recommended treatment options.
Tumours that show local invasion (T4) or distant metastases (M1) are not amenable to curative treatment.
Curative treatment
Patients deemed medically fit with non-metastatic or locally invasive tumours should be offered surgical resection to cure early cancers (Stage I-IIA) and chemotherapy (neoadjuvant) followed by surgical resection for higher stage tumours (Stage IIB+) as it improves long-term survival.16
An oesophagectomy is performed by either an abdominal incision and transhiatal approach (to mobilise the stomach and a subsequent neck incision to pull up the stomach into the mediastinum and remove the oesophagus) or as a transthoracic Ivor-Lewis Oesophago-gastrectomy which involves an abdominal incision and a left-sided thoracotomy.
Both methods have been found to have similar hospital mortality and five-year survival rates. This highly invasive surgery is associated with significant morbidity and complications (30%), hospital mortality (2.9%)30 and reduction in long-term quality of life31 therefore results of surgery are subject to national audit30 and performed in centres with higher case volumes to achieve better results.32
Multiple meta-analyses have shown the benefits of preoperative (neoadjuvant) chemotherapy or radiotherapy in patients undergoing surgery. Two cycles of neoadjuvant chemotherapy have been shown to improve survival over two years from 34 to 43% without additional serious adverse events in a large UK Medical Research Council study. This effect is notable especially for patients with T3 disease or the presence of lymph nodes and is therefore used in most UK centres.33-35
Given its response to radiotherapy, definitive (curative) chemoradiotherapy can be an option for localised OSCC (i.e. all areas within a radiation field) especially if it is affecting the upper oesophagus. Although surgery seems to be a better option in comparison,36 some studies have shown equivalent two-year survival to surgery in this group, therefore chemoradiotherapy remains a recommended first-line option for OSCC.37,16
Advances in endoscopic imaging now result in detection of early,non-ulcerating carcinomas at screening before dysphagia develops. In small nodular lesions < 2 cm, endoscopic mucosal resection may be considered to stage and treat early cancers and differentiates between high-grade dysplasia, T1a and T1b lesions. Endoscopic removal may be complete and considered curative in T1a given the low incidence of lymph node metastases in this group (< 5%), and avoids surgery.
Palliative treatment
Most patients presenting with oesophageal cancer have incurable metastases at diagnosis. A palliative treatment plan should be considered by the MDT, taking into account performance status and patient preference. Early direct involvement of the palliative care team, the cancer nurse specialists and dieticians (all core members of the MDT) is essential.
Palliative combination chemotherapy can be offered in advanced oesophageal cancer. Trials have shown response to palliative chemotherapy in 37-48% of patients. Mean survival ranges from 8 to 13 months with better outcomes in OSCC groups.38,39 In patients with advanced OAC involving the upper stomach, endoscopic biopsies are assessed for HER-2 immunopositivity. The addition of trastuzumab can result in a statistically significant improvement in response rate and median overall survival (13.8 versus 11.1 months) in patients with HER-2 receptive tumours.40,41
Dysphagia is the predominant symptom in patients with oesophageal cancer. Self-expanding metal stents (SEMS) can be used to aid dysphagia and nutrition. They can be placed endoscopically or radiologically in a single procedure.16 When SEMS are compared with other methods to help swallowing, such as endoscopy with argon photocoagulation debulking, they have similar outcomes on quality of life, but debulking requires multiple procedures so is avoided in those patients with limited life expectancy.42 Complications of SEMS are stent migration, pain for up to ten days, blockage and stent overgrowth by tumour requiring further stents or endoscopy in one third of cases.43 Dilatation is rarely used because of the high risk of perforation and early recurrence and percutaneous endoscopic gastrostomy placement is only rarely used.
support and follow-up
Dietician review and cancer nurse specialist input has been demonstrated to contribute to improved quality of life.44 The cancer nurse specialist is central to patient care, consulting with multiple specialties including primary care to provide a co-ordinated approach and act as the patient’s advocate.45
Regular review of patients following therapy is required to manage post-treatment side effects such as dysphagia and post-surgical diarrhoea and pain.
Regular access to cancer nurse specialists has been shown to be cost effective in supporting follow-up.46
In the palliative setting these nurses can ensure close liaison with primary and secondary care and help avoid readmission for relief of pain, nutrition and dysphagia.47
Future directions
The assessment and evaluation of outcomes is fundamental in the management of oesophageal cancer. The National Oesophago-Gastric
Cancer Audit has set high standards.30 With drivers like this it is encouraging to see cancer registries across Europe reporting gradual improvements in
five-year survival rates, however, they are still generally poor and varied.
The observed trends reflect the variations in alcohol consumption, smoking and obesity across European countries.48 With the incidence remaining high we need to develop effective treatments with limited morbidity that minimise significant effects on quality of life and health service resources. Minimal access surgery and developments in endoscopy are encouraging.49
Preventative strategies to improve rates of oesophageal cancer including smoking cessation and weight reduction are required in the UK. Increasing expertise and evidence for treating early lesions in Barrett's oesophagus mean less invasive endoscopic techniques can be used to prevent cancer development.50
Given the advanced stage of oesophageal cancer at presentation, waiting for patients to develop alarm symptoms before referring them for endoscopy would seem not to improve our five-year survival rates. There are currently very limited non-invasive biomarkers to detect or screen for early oesophageal cancer. Research to develop a novel approach to early diagnosis of Barrett’s oesophagus and dysplasia using a swallowed cell collection device (Cytosponge), coupled with molecular assays is in development and may hold a possible option for future screening.53
In the meantime oesophageal cancer still has one of the lowest cancer survival outcomes in the UK and a low threshold for early endoscopy for dysphagic symptoms is recommended.
Further reading
Allum WH, Blazeby JM, Griffin SM et al. On behalf of the Association of Upper Gastrointestinal Surgeons of Great Britain and Ireland, the British Society of Gastroenterology and the British Association of Surgical Oncology. Guidelines for the management of oesophageal and gastric cancer. Gut 2011;60:1449-72
Scottish Intercollegiate Guidelines Network. SIGN 87. Management of oesophageal and gastric cancer. SIGN. Edinburgh. 2006 www.sign.ac.uk/pdf/sign87.pdf
National Institute for Health and Care Excellence. NG12. Referral guidelines for suspected cancer. NICE. London. 2015 www.nice.org.uk/guidance/ng12
National Oesophago-gastric Cancer Audit - 2013, Annual report. 2013 www.hscic.gov.uk/catalogue/PUB11093/clin-audi-supp-prog-oeso-gast-2013-rep.pdf
REFERENCES
References
1 Office for National Statistics. Cancer Registration Statistics England 2013. 10 July 2015. www.ons.gov.uk/ons/dcp171778_409714.pdf
2 Office for National Statistics. Cancer Survival in England: adults diagnosed 2009 to 2013 Followed up to 2014. 19 November 2015 www.ons.gov.uk/ons/dcp171778_424443.pdf
3 Cancer Research UK: Oesophageal cancer statistics. www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/oesophageal-cancer#heading-. Accessed January 2016.
4 Chung CS, Lee YC, Wang CP et al. Secondary prevention of esophageal squamous cell carcinoma in areas where smoking, alcohol, and betel quid chewing are prevalent. J Formos Med Assoc 2010;109:408-21
5 Lagergren J, Bergstrom R, Lindgren A, Nyren O. Symptomatic gastroesophageal reflux as a risk factor for esophageal adenocarcinoma. N Engl J Med 1999;340:825-31
6 Steffen A, Huerta JM, Weiderpass E et al. General and abdominal obesity and risk of esophageal and gastric adenocarcinoma in the European Prospective Investigation into Cancer and Nutrition. Int J Cancer 2015;137:646-57
7 Bhat S, Coleman HG, Yousef F et al. Risk of malignant progression in Barrett's esophagus patients: results from a large population-based study. J Natl Cancer Inst 2011;103:1049-57
8 Hvid-Jensen F, Pedersen L, Drewes AM et al. Incidence of adenocarcinoma among patients with Barrett's esophagus. N Engl J Med 2011;365:1375-83
9 Fitzgerald RC, di Pietro M, Ragunath K et al. British Society of Gastroenterology guidelines on the diagnosis and management of Barrett's oesophagus. Gut 2014;63:7-42
10 Hopper AD. Improving the diagnosis and management of GORD in adults. Practitioner 2015;259:27-32
11 Kapoor N, Bassi A, Sturgess R, Bodger K. Predictive value of alarm features in a rapid access upper gastrointestinal cancer service. Gut 2005;54:40-5
12 Wong CJ. Involuntary weight loss. Med Clin North Am 2014;98:625-43
13 Gaddey HL, Holder K. Unintentional weight loss in older adults. Am Fam Physician 2014;89:718-22
14 National Institute for Health and Care Excellence. NG12. Referral guidelines for suspected cancer. NICE. London. 2015. www.nice.org.uk/guidance/ng12
15 Scottish Intercollegiate Guidelines Network. SIGN 87. Management of oesophageal and gastric cancer. SIGN. Edinburgh. 2006 www.sign.ac.uk/pdf/sign87.pdf
16 Allum WH, Blazeby JM, Griffin SM et al. Guidelines for the management of oesophageal and gastric cancer. Gut 2011;60:1449-72
17 Yalamarthi S, Witherspoon P, McCole D, Auld CD. Missed diagnoses in patients with upper gastrointestinal cancers. Endoscopy 2004;36:874-9
18 Bramble MG, Suvakovic Z, Hungin AP. Detection of upper gastrointestinal cancer in patients taking antisecretory therapy prior to gastroscopy. Gut 2000;46:464-7
19 Levine MS, Chu P, Furth EE et al. Carcinoma of the esophagus and esophagogastric junction: sensitivity of radiographic diagnosis. AJR Am J Roentgenol 1997;168:1423-6
20 Rice TW, Ishwaran H, Blackstone EH. Oesophageal cancer: location, location, location. Eur J Cardiothorac Surg 2015;48:194-5
21 Kuszyk BS, Bluemke DA, Urban BA et al. Portal-phase contrast-enhanced helical CT for the detection of malignant hepatic tumors: sensitivity based on comparison with intraoperative and pathologic findings. AJR Am J Roentgenol 1996;166:91-5
22 Picus D, Balfe DM, Koehler RE et al. Computed tomography in the staging of esophageal carcinoma. Radiology 1983;146:433-8
23 Umeoka S, Koyama T, Watanabe G et al. Preoperative local staging of esophageal carcinoma using dual-phase contrast-enhanced imaging with multi-detector row computed tomography: value of the arterial phase images. J Comput Assist Tomogr 2010;34:406-12
24 Murray P, Whiting P, Hutchinson SP et al. Preoperative shuttle walking testing and outcome after oesophagogastrectomy. Br J Anaesth 2007;99:809-11
25 Feeney C, Hussey J, Carey M, Reynolds JV. Assessment of physical fitness for esophageal surgery, and targeting interventions to optimize outcomes. Dis Esophagus 2010;23:529-39
26 Pech O, Gunter E, Dusemund F et al. Accuracy of endoscopic ultrasound in preoperative staging of esophageal cancer: results from a referral center for early esophageal cancer. Endoscopy 2010;42:456-61
27 Meyers BF, Downey RJ, Decker PA et al. The utility of positron emission tomography in staging of potentially operable carcinoma of the thoracic esophagus: results of the American College of Surgeons Oncology Group Z0060 trial. J Thorac Cardiovasc Surg 2007;133:738-45
28 de Graaf GW, Ayantunde AA, Parsons SL et al. The role of staging laparoscopy in oesophagogastric cancers. Eur J Surg Oncol 2007;33:988-92
29 Washington K. 7th edition of the AJCC cancer staging manual: stomach. Ann Surg Oncol 2010;17:3077-79
30 National Oesophago-gastric Cancer Audit - 2013, Annual report: June 26, 2013. www.hscic.gov.uk/catalogue/PUB11093/clin-audi-supp-prog-oeso-gast-2013-rep.pdf
31 Djarv T, Lagergren J, Blazeby JM, Lagergren P. Long-term health-related quality of life following surgery for oesophageal cancer. Br J Surg 2008;95:1121-26
32 Bachmann MO, Alderson D, Edwards D et al. Cohort study in South and West England of the influence of specialization on the management and outcome of patients with oesophageal and gastric cancers. Br J Surg 2002;89:914-22
33 Medical Research Council Oesophageal Cancer Working Group. Surgical resection with or without preoperative chemotherapy in oesophageal cancer: a randomised controlled trial. Lancet 2002;359:1727-33
34 Cunningham D, Allum WH, Stenning SP et al. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N Engl J Med 2006;355:11-20
35 Sjoquist KM, Burmeister BH, Smithers BM et al. Survival after neoadjuvant chemotherapy or chemoradiotherapy for resectable oesophageal carcinoma: an updated meta-analysis. Lancet Oncol 2011;12:681-92
36 Stahl M, Stuschke M, Lehmann N et al. Chemoradiation with and without surgery in patients with locally advanced squamous cell carcinoma of the esophagus. J Clin Oncol 2005;23:2310-7
37 Bedenne L, Michel P, Bouche O et al. Chemoradiation followed by surgery compared with chemoradiation alone in squamous cancer of the esophagus: FFCD 9102. J Clin Oncol 2007;25:1160-08
38 Van Cutsem E, Moiseyenko VM, Tjulandin S et al. Phase III study of docetaxel and cisplatin plus fluorouracil compared with cisplatin and fluorouracil as first-line therapy for advanced gastric cancer: a report of the V325 Study Group. J Clin Oncol 2006;24:4991-97
39 Cunningham D, Starling N, Rao S et al. Capecitabine and oxaliplatin for advanced esophagogastric cancer. N Engl J Med 2008;358:36-46
40 Bang YJ, Van Cutsem E, Feyereislova A et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet 2010;376:687-97
41 Rivera F, Carrato A, Gravalos C et al. Recommendations on current approach to gastric cancer. Clin Transl Oncol 2009;11:518-25
42 Diamantis G, Scarpa M, Bocus P et al. Quality of life in patients with esophageal stenting for the palliation of malignant dysphagia. World J Gastroenterol 2011;17:144-50
43 Mangiavillano B, Pagano N, Arena M et al. Role of stenting in gastrointestinal benign and malignant diseases. World J Gastrointest Endosc 2015;7:460-80
44 Isenring EA, Capra S, Bauer JD. Nutrition intervention is beneficial in oncology outpatients receiving radiotherapy to the gastrointestinal or head and neck area. Br J Cancer 2004;91:447-52
45 Douglas HR, Halliday D, Normand C et al. Economic evaluation of specialist cancer and palliative nursing: Macmillan evaluation study findings. Int J Palliat Nurs 2003;9:429-38
46 Verschuur EM, Steyerberg EW, Tilanus HW et al. Nurse-led follow-up of patients after oesophageal or gastric cardia cancer surgery: a randomised trial. Br J Cancer 2009;100:70-6
47 Leary A, Crouch H, Lezard A et al. Dimensions of clinical nurse specialist work in the UK. Nurs Stand 2008;23:40-4
48 Castro C, Bosetti C, Malvezzi M et al. Patterns and trends in esophageal cancer mortality and incidence in Europe (1980-2011) and predictions to 2015. Ann Oncol 2014;25:283-90
49 Safranek PM, Cubitt J, Booth MI, Dehn TC. Review of open and minimal access approaches to oesophagectomy for cancer. Br J Surg 2010;97:1845-53
50 Patel V, Burbridge RA. Endoscopic approaches for early-stage esophageal cancer: current options. Curr Oncol Rep 2015;17:421
51 Abdel Jalil AA, Katzka DA, Castell DO. Approach to the patient with dysphagia. Am J Med 2015;128:1138 e17-23
52 Rice TW. Esophageal Cancer Staging. Korean J Thorac Cardiovasc Surg 2015;48:157-63
53 Fitzgerald RC. Combining simple patient-oriented tests with state-of-the-art molecular diagnostics for early diagnosis of cancer. United European Gastroenterol J 2015;3:226-9
ARTICLE IN PDF
PRESET SEARCHES
PubMed
NICE Evidence search
US National Institute of Health
Immediate CPD prompts and frameworks
These handy templates* prompt immediate drafting of personal learning, reflection and planning. Save your record straight into your personal development folder on your device or Cloud storage system.
*Adobe reader needed for iPad

 = Registered users
= Registered users = Paid-up subscribers
= Paid-up subscribers