Preset searches
PubMed -Respiratory medicine
Obstructive sleep apnoea hypopnoea
Tuberculosis primary care review
Upper respiratory tract infections
Lower respiratory tract infections
Shepherd JG, Chapman ALN. Assessment and management of active and latent TB. Practitioner 2016;260(1798)21-24
Assessment and management of active and latent TB
24 Nov 2016
AUTHORS
Dr James G Shepherd, BSc (Hons) MBChB DTM&H MRCP, Specialty Registrar in Infectious Diseases and Medical Microbiology, West of Scotland Deanery, UK
Dr Ann LN Chapman BM BCh FRCP DTM&H MSc PhD, Consultant in Infectious Diseases and General Medicine, NHS Lanarkshire, UK
Article
Abstract
More than 25% of the world population has been infected with tuberculosis (TB), however only 10% of those infected will ever develop active disease. Clinically significant disease occurs through progression of primary infection or through later reactivation of latent TB infection (LTBI); this is most likely to occur in the first few years following infection, although late reactivation can occur several decades later, particularly in individuals who become immunosuppressed. Risk of TB acquisition is increased in people who have come to the UK from high incidence countries or who are born in the UK but come from high-risk ethnic minority groups. In 2015, 73% of those diagnosed with active TB were born outside the UK. Other risk groups include those who are homeless, in prison or who misuse drugs or alcohol. Once infected people who are immunosuppressed are at greater risk of progression to active disease. Infants below the age of 12 months can develop rapidly progressive and potentially fatal infection. Initial clinical assessment with chest radiography and the collection of three deep respiratory samples for smear microscopy and culture remain the standard of care. The management of active TB has not changed significantly over many years. The most significant changes in the 2016 NICE guidance relate to screening for LTBI in individuals who are contacts of a patient with active TB, or who are recent entrants to the UK from a high incidence country. NICE recommends that only contacts of patients with active pulmonary or laryngeal TB be screened.
Article
Tuberculosis (TB) is one of the leading global causes of mortality.1 It is estimated that more than 25% of the world population has been infected, however only 10% of infected individuals will ever develop active disease.2
In the majority of exposed individuals, the immune system either eradicates the infection in its early stages or achieves a degree of containment, resulting in the bacterium entering a dormant state known as latent TB infection (LTBI). Active infection resulting in clinically significant disease occurs through progression of primary infection or through later reactivation of LTBI; this is most likely to occur in the first few years following infection, although late reactivation can occur several decades later, particularly in individuals who become immunosuppressed through disease or therapy.
There were an estimated 10.4 million new TB cases globally in 2015. Worldwide incidence and mortality have steadily fallen since 2003.3 Although the UK is a low incidence country with a robust healthcare infrastructure, TB notifications in England and Wales rose in the first decade of the 21st century, reaching a peak of 8,371 in 2011 before falling to 5,859 in 2015.4,5 The bulk of cases are among migrants: 73% of those diagnosed with active TB in 2015 were born outside the UK.
UK incidence remains significantly higher than similar developed countries, prompting a reorganisation of national TB control as set out in Public Health England’s Collaborative Tuberculosis Strategy for England 2015.6 NICE published an updated guideline on TB earlier this year.7 One aim of the 2016 guideline was to provide a comprehensive resource for TB services incorporating all the areas previously covered in NICE clinical and public health guidelines.
TB is spread through inhalation of respiratory droplets from patients with active pulmonary or laryngeal TB; infection can only be transmitted if patients are producing sputum.
It is important to consider the risk of both acquisition of TB and also of progression to active infection.
Risk of acquisition is increased in people who have come to the UK from high incidence countries or who are born in the UK but come from high-risk ethnic minority groups. Other risk groups in the UK include those who are homeless, in prison or who misuse drugs or alcohol. Once infected, people who are immunosuppressed are at greater risk of progression to active disease, see table 1. One group at particularly high risk are infants below the age of 12 months who can develop rapidly progressive and potentially fatal infection. For this reason, it is important for TB teams to be aware of infants who may have been exposed to an infectious case and to refer the child to paediatric services as a matter of urgency.
Active TB
Assessment of active disease
A clear distinction should be drawn between the investigation of suspected active TB and the detection of LTBI, as the diagnostic and therapeutic pathways differ significantly.
The presenting symptoms of active TB are common in primary care and as such a high degree of suspicion is necessary to make the diagnosis. Cough is the main symptom in pulmonary disease but is not always accompanied by sputum production. Malaise, weight loss, fever and night sweats are common to both pulmonary and extrapulmonary disease. The presence of such symptoms, particularly in a patient with risk factors for TB, should prompt further investigation.
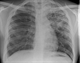
For suspected pulmonary disease the initial clinical assessment has not changed in decades, with chest radiography and the collection of three deep respiratory samples for smear microscopy and culture remaining the standard of care, see figure 1.
If the patient is suspected of having active TB but is unable to produce a sputum sample, GPs should refer the patient to their local TB team for further assessment, which may include an induced sputum procedure or bronchoscopy.
Recent laboratory advances have improved the sensitivity and speed of TB diagnosis, for example the use of fluorescence microscopy, rather than traditional Ziehl-Neelsen staining, for sputum smear examination.8
Additionally, molecular diagnostic tests known as nucleic acid amplification tests (NAATs) can be performed directly on sputum, produce rapid results, and have a significantly higher sensitivity than smear microscopy. Some NAATs are able to detect the presence of genes signifying rifampicin resistance, a valuable tool in the assessment of potentially multidrug-resistant disease.9 However, in view of their cost NAAT assays are recommended only in situations where rapid confirmation of TB and/or rifampicin sensitivity is important, for example where there may be a need for a large contact tracing exercise or where multidrug resistance is suspected.
Management of active disease
The management of active TB has not changed significantly over many years.10 Attempts to shorten treatment courses using novel agents such as fluoroquinolones have not yet been successful.11
The standard regimen has two stages: a two-month initiation phase involving a four-drug combination of rifampicin, isoniazid, pyrazinamide and ethambutol, followed by a continuation phase of rifampicin and isoniazid lasting at least four months. In the initiation stage ethambutol is included in case of drug resistance and can be discontinued early once there is confirmation of fully sensitive disease. Fixed dose combination tablets are available for both the initiation and continuation phases, and their use is recommended by NICE to aid compliance and reduce pill burden.
Although the standard regimen is generally well tolerated it can be associated with serious side effects. Isoniazid, rifampicin and pyrazinamide have the potential to induce hepatotoxicity, and patients should be warned of this and advised to report symptoms such as nausea, vomiting or jaundice. Liver enzymes should be measured before starting treatment, however serial monitoring on therapy is only indicated if there are abnormalities at baseline.
Ethambutol can cause optic neuritis so visual acuity should be measured before starting therapy and the patient should contact the TB team or their GP if they become aware of any changes in their vision. Symptoms such as rashes and nausea are more common but usually respond to symptomatic measures.
Latent TB infection (LTBI)
Assessment of latent disease
The most significant changes in the 2016 NICE guidance relate to recommendations on screening for LTBI in individuals who are contacts of a patient with active TB, or who are recent entrants to the UK from a high incidence country, see table 2.
The tuberculin skin test (TST) in the form of the Heaf (now discontinued) or Mantoux test has been used for decades for the detection of LTBI and was the only test available until the introduction of the interferon gamma release assays (IGRAs) in the mid 2000s.
IGRAs are blood based in vitro tests that measure T cell responses to specific antigens of M. tuberculosis. As with the TST, a positive reaction indicates immunological memory of mycobacterial antigens and acts as a surrogate marker for LTBI. IGRAs are more specific than Mantoux and are not affected by prior BCG vaccination or exposure to most non-tuberculous mycobacteria.12 Additionally, they are not subject to the logistical constraints of TST, with results potentially available within 24 hours and no requirement for a follow-up appointment for results to be interpreted.
The drawback of IGRAs is their high cost relative to Mantoux. As such, NICE had previously recommended a strategy of an initial Mantoux screening test followed by a confirmatory IGRA in most circumstances. However, the recent guidance has returned to predominant use of the TST on grounds of cost effectiveness. In addition, the evidence considered by the NICE guideline development group supported a change in the cut-off between a positive and negative Mantoux test, such that a positive result is now defined as an induration of 5 mm or greater irrespective of prior BCG vaccination.
Previously it was recommended that household contacts of any patient with active TB should be screened for latent infection. The recent guidance recommends that only contacts of patients with active pulmonary or laryngeal TB should be screened in order to focus resources on contacts of the most highly infectious cases.
Although this approach will in theory result in fewer contacts being screened with a higher proportion of contacts being found to have LTBI, one potential disadvantage is that we lose the opportunity to identify people with LTBI who may not have acquired infection from the non-infectious index case in question, but from a third party acting as a common source. Conversely, the reduction in the Mantoux threshold, and increase in age cut-off for screening, may increase screening workload.
It will be important to assess the impact of these changes on future TB epidemiology in the UK.
Management of latent disease
Standard treatment for LTBI is with three months of rifampicin and isoniazid, or six months of isoniazid alone. The former regimen is associated with reduced hepatotoxicity and improved compliance. The isoniazid only regimen is of value in patients where there is risk of drug interactions with rifampicin: important interactions occur with oral or implanted contraceptives, warfarin and methadone, among many other drugs.
One further significant change in LTBI management relates to the age cut-off for treatment of LTBI. In previous guidance this cut-off was 35 years, based on increasing risk of hepatotoxicity and reducing efficacy with increasing age. Notably, the upper age limit for offering to test and treat individuals for LTBI is now 65 years, with the caveat that individuals aged 35 to 65 years should only be treated if there is no concern about hepatotoxicity.
Conclusions
The expansion in the use of immunosuppressive medications exposes more individuals to the risk of reactivation of LTBI. As such it is important that healthcare professionals in frontline services are aware of the local epidemiology and common presenting symptoms of TB.
Clear referral pathways from GP and emergency departments are required to ensure possible cases of active TB are assessed rapidly and treatment started as soon as possible to minimise clinical severity and public health impact.
Despite developments in novel diagnostics it is important to bear in mind that sputum samples and chest radiography remain the mainstay of diagnosis for suspected active pulmonary disease.
REFERENCES
1 GBD 2015 Mortality and Causes of Death Collaborators. Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980–2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet 2016;388:1459-1544
2 Vynnycky E, Fine PE. The natural history of tuberculosis: the implications of age dependent risks of disease and the role of reinfection. Epidemiol Infect 1997;119:183-201
3 World Health Organization. Global Tuberculosis Report 2016. WHO. Geneva. 2016 www.who.int/tb/publications/global_report/en/
4 Public Health England. TB case notifications, England and Wales 1913-2015 www.gov.uk/government/publications/tuberculosis-tb-annual-notifications-1913-onwards
5 Public Health England. Tuberculosis in England 2016 report. 2016 PHE publications
www.gov.uk/government/publications/tuberculosis-in-england-annual-report
6 Public Health England. Collaborative tuberculosis strategy for England 2015 to 2020. 2015 PHE publications
www.gov.uk/government/publications/collaborative-tuberculosis-strategy-for-england
7 National Institute for Health and Care Excellence. NG33. Tuberculosis. NICE. London. 2016 www.nice.org.uk/guidance/NG33/chapter/Recommendations
8 Steingart KR, Henry M, Ng V et al. Fluorescence versus conventional sputum smear microscopy for tuberculosis: a systematic review. Lancet Infect Dis 2006;6(9):570-81
9 Steingart KR, Schiller I, Horne DJ et al. Xpert® MTB/RIF assay for pulmonary tuberculosis and rifampicin resistance in adults. Cochrane Database Syst Rev 2014, Issue 1. Art. No.: CD009593
10 Hong Kong Chest Service/British Medical Research Council. Controlled trial of 2, 4, and 6 months of pyrazinamide in 6-month, three-times-weekly regimens for smear-positive pulmonary tuberculosis, including an assessment of a combined preparation of isoniazid, rifampin, and pyrazinamide. Results at 30 months. Am Rev Respir Dis 1991;143(4 Pt 1):700-06
11 Gillespie SH, Crook AM, McHugh TD et el. Four-month moxifloxacin-based regimens for drug-sensitive tuberculosis. N Engl J Med 2014;371(17):1577-87
12 Pai M, Denkinger CM, Kik SV et al. Gamma interferon release assays for detection of Mycobacterium tuberculosis infection. Clin Microbiol Rev 2014;27(1):3-20
ARTICLE IN PDF
PRESET SEARCHES
Pubmed
Tuberculosis primary care review
NICE evidence
Immediate CPD prompts
These handy templates*, associated with all articles, prompt drafting of personal learning, reflection and planning.
Save your record straight into your personal development folder on your device or cloud storage system.
*Adobe fillable PDFs. Adobe reader needed for iPad
Comment or question
If you, or your peer group, would like to comment on an article or have a question for the authors, write to:
Editor
Case report submissions
Do you have an interesting case that you would like to share with other readers?
Your article would need to be around 1,500 words long, include a brief review of the literature to put the case in context, and be fully referenced in the Vancouver style.
Submissions for consideration for publication should be sent to: [email protected]

 = Registered users
= Registered users = Paid-up subscribers
= Paid-up subscribers