Hackett S, Mahmood F, Coates L, Helliwell P. Prompt diagnosis key in improving outcomes in psoriatic arthritis. Practitioner October 2022;266(1862):17-21
Prompt diagnosis key in improving outcomes in psoriatic arthritis
25 Oct 2022
AUTHORS
Dr Simon Hackett BSc BM BS MSc DPhil, Research Fellow in Rheumatology, Nuffield Department of Orthopaedics, Rheumatology and Musculoskeletal Sciences, University of Oxford, Oxford, UK
Dr Farrouq Mahmood MBChB, Clinical Research Fellow in Rheumatology, Bradford Teaching Hospitals NHS Trust, Bradford, UK
Dr Laura Coates MBChB PhD, Associate Professor in Rheumatology, Nuffield Department of Orthopaedics, Rheumatology and Musculoskeletal Sciences, University of Oxford, Oxford, UK
Professor Philip Helliwell MA DM PhD FRCP, Professor of Clinical Rheumatology, Leeds Institute of Rheumatic and Musculoskeletal Medicine, University of Leeds, Leeds, UK
Article
If left untreated psoriatic arthritis (PsA) can result in progressive, irreversible joint damage causing chronic pain and disability. It is a chronic, autoimmune inflammatory condition that can affect up to 30% of patients with psoriasis. PsA is a multisystem disease affecting other organs such as the eyes, gut and tendons. It is associated with multiple comorbidities such as diabetes mellitus, cardiovascular disease and metabolic syndrome. Early diagnosis is crucial as structural joint damage can occur within the first two years of disease onset. Symptoms in PsA patients have traditionally been divided into six domains: peripheral arthritis, enthesitis, dactylitis, axial disease, psoriasis and nail disease. Patients do not need to have symptoms in all domains for diagnosis, but examination of these six domains is important when assessing patients, prior to making a referral to rheumatology. If PsA is suspected in primary care, routine blood tests should be requested to check for anaemia of chronic disease and raised inflammatory markers. Plain radiographs of hands and feet should be requested prior to referral, although classical changes do not occur early in the disease. If GPs suspect PsA, prompt referral is paramount. Patients should be referred if they have persistent joint symptoms, a positive screening questionnaire (PEST), joint swelling, dactylitis or enthesitis, and/or inflammatory back pain.
If left untreated psoriatic arthritis (PsA) can result in progressive, irreversible joint damage causing chronic pain and disability.
It is a chronic, autoimmune inflammatory condition that can affect up to 30% of patients with psoriasis.1 PsA is a multisystem disease affecting other organs such as the eyes, gut and tendons. Furthermore, it is associated with multiple comorbidities such as diabetes mellitus, cardiovascular disease and metabolic syndrome.2 It is increasingly recognised that PsA has a severe burden of disease for patients which affects day to day life. Quality of life can be severely affected with some patients unable to work, and there can be associated depression and anxiety.2
There is evidence that early diagnosis and treatment can result in better outcomes. Indeed, early diagnosis is crucial as structural joint damage can occur within the first two years of disease onset.2 PsA presentation is often clinically heterogeneous; symptoms and signs may also overlap with other musculoskeletal conditions such as osteoarthritis (OA) and rheumatoid arthritis (RA), increasing the risk of misdiagnosis.1 Early identification and referral of patients with a clinical suspicion of PsA by GPs is key to ensure optimal diagnosis and management.
Clinical presentations
Although most patients will usually have a history of psoriasis, in 15% of cases arthritis can precede skin features.2
There is no diagnostic test for PsA, patients are diagnosed based on clinical symptoms and signs. Typically, tests for rheumatoid factor (RF) and anti-cyclic citrullinated peptide (anti-CCP) are negative and up to 50% of those with active arthritis will have a normal C-reactive protein (CRP). Symptoms in PsA patients have traditionally been divided into domains which are: peripheral arthritis, enthesitis, dactylitis, axial disease, psoriasis and nail disease. Patients do not need to have symptoms in all domains to be diagnosed, but examination of these six domains is important when assessing patients in primary care, prior to making a referral to a rheumatologist.
Peripheral arthritis
Although PsA can present as a symmetrical polyarthritis similar to RA, it can also cause a monoarthritis or oligoarthritis associated with early morning stiffness lasting longer than 30 minutes. Joint pain is often worse at rest and improves with activity. Joint swelling is an important symptom and clinical sign.
Dactylitis
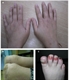
Diffuse swelling of an entire digit, dactylitis, can occur in up to half of PsA patients, most commonly the index finger and the fourth toe.1 Dactylitis can present as a tender, often erythematous, warm digit or as a swollen, asymptomatic digit: it is commonly known as a ‘sausage’ digit (see figure 1A).
Enthesitis
Inflammation of the enthesis, commonly affecting the Achilles tendon insertion at the heel, can also be a presenting feature. Patients should be assessed clinically for signs of enthesitis (see figure 1B).
Psoriasis
Skin inflammation is common in PsA patients and up to a third of patients with psoriasis subsequently develop PsA. Cutaneous risk factors are scalp and nail involvement, and psoriasis in the flexures such as the natal cleft, the groin and under the breasts. During clinical examination areas such as the elbows, knees, natal cleft and scalp should be examined for any signs of psoriasis. Patients with pre-existing psoriasis should be regularly screened for PsA using a simple tool such as the Psoriasis Epidemiology Screening Tool (PEST) every six months (see figure 2). Regular monitoring and follow-up of patients with psoriasis to monitor for joint symptoms should be carried out in primary care, with regular reminders set in patient records.
Nail disease
The presence of psoriatic nail dystrophy, including pitting, leukonychia, discoloration and onycholysis, is associated with PsA.4 The nails of both hands and feet should be examined in patients to assess for any signs of nail disease (see figure 1C).
Axial disease
Between 25 and 70% of patients with PsA have evidence of axial disease, with some patients exclusively experiencing axial joint involvement. Axial disease is characterised by inflammation and post-inflammatory structural changes of the spine and/or sacroiliac joints.
Patients may describe symptoms consistent with inflammatory back pain indicating sacroiliitis:6
• Onset before the age of 40
• Gradual onset
• Nocturnal pain
• Improvement with activity
• No improvement with rest
Given the heterogeneous nature of PsA, studies are underway to develop clinical scoring systems to enable clinicians to make an early clinical diagnosis of PsA. The new scoring systems will incorporate both historical, clinical and imaging findings. It is also hoped that a simple blood test will eventually be developed. The CASPAR criteria can be applied in a clinical setting but only in patients with inflammation of joints, spine or entheses5 (see table 1). These criteria have been shown to be more sensitive than other criteria for classifying early PsA.
Investigations
If a diagnosis of PsA is suspected in primary care, routine blood tests should be requested to check for anaemia of chronic disease and raised inflammatory markers, although it should be noted that CRP is raised in only 50% of patients,2 thus a normal CRP does not exclude the diagnosis, but if present could predict a more severe disease course. PsA patients are usually negative for RF and CCP antibodies; the presence of these autoantibodies may help distinguish between RA and PsA.
Plain radiographs of hands and feet should be requested prior to referral, although classical changes do not occur early in the disease. PsA has typical appearances on X-ray which can aid diagnosis although normal X-rays will be common and should not delay a referral. X-rays may show peripheral joint erosions with fluffy margins along the bones, periostitis, indicating new bone formation.
Typically, the distal interphalangeal (DIP) joints are affected in PsA; in RA erosions are more likely to affect the metacarpophalangeal (MCP) and proximal interphalangeal (PIP) joints in a symmetrical fashion. Patients with severe disease may have osteolysis of the joint, pencil-in-cup deformity. Plain films of the sacroiliac joints may show sacroiliitis which can be either unilateral or bilateral in inflammatory back pain.
X-rays may be normal in early disease. In secondary care ultrasound is increasingly used to help identify inflammatory arthritis in difficult cases. As well as erosions, it can identify enthesitis and soft tissue inflammation which can be difficult to recognise on plain films. Ultrasound may also be useful in obese patients where it is difficult to assess for synovitis or dactylitis clinically.7
Referral
If GPs suspect PsA, prompt referral is paramount. Patients should be referred if they have persistent joint symptoms (lasting more than 3 months), a positive screening questionnaire (PEST), joint swelling, dactylitis or enthesitis, and/or inflammatory back pain. In recent years, the proportion of people with suspected persistent joint inflammation that have been referred to rheumatology services within three working days of GP contact has increased. Although this is encouraging, many PsA patients still experience a diagnostic delay.
Recent evidence from the Swedish Early Psoriatic Arthritis Register (SwePsA) demonstrated that in early PsA, a short delay between onset of symptoms and diagnosis, preserved function, and male gender are the most important predictors of long-term favourable clinical outcomes.14
Considering PsA as a potential diagnosis in patients presenting with inflammatory arthritis symptoms may help reduce potential delays.
Treatment
Patients may experience problems with their joints and skin concurrently and the clinical heterogeneity of PsA makes it a complex disease to manage. Patients on PsA treatment regimens require regular blood monitoring.
Side effects may lead to lack of compliance. Physicians may have concerns over long-term tolerability and safety of systemic therapies leading to undertreatment of the disease.8
For those who present with mild symptoms of joint inflammation and low disease activity, NSAIDs can be used for symptom relief. Agents such as naproxen can be prescribed while the patient is waiting for specialist assessment. The usual precautions should be observed when prescribing NSAIDs.
Steroid therapy can also be used for symptom relief in more severe cases, either as a short course of oral prednisolone or as an intramuscular injection. Patients should be warned that there is a risk of psoriasis flare with both NSAID and steroid use.2 Steroids could also mask symptoms and signs when a patient is being assessed by a specialist, thus leading to a delay in a definitive diagnosis.
Patients should be referred to physiotherapy to help with exercise and rehabilitation, and occupational therapy if needed. This is usually done by secondary care. Counselling should be an option for patients as chronic pain and disability can be associated with low mood and anxiety.
Treat to target
Guidance issued earlier in 2022 by the British Society for Rheumatology (BSR) has redefined the treatment algorithm for PsA.15 Importantly, the concept of treat to target, using regular review and escalation of therapy according to a prespecified objective target, has been applied in the context of the proposed new treatment algorithm.15
Disease modifying antirheumatic drugs (DMARDs): First- and second-line treatment of PsA is normally with conventional systemic DMARDs (csDMARD) such as methotrexate, sulfasalazine and leflunomide. Methotrexate is typically used as a first-line DMARD as it has been shown to improve both skin and joint problems. Clinicians should be vigilant for side effects such as nausea, leucopenia, abnormal liver function tests and, pneumonitis (though rare), and regular blood tests are required.
Patients who respond inadequately to csDMARDs are often started in secondary care on biologic drugs.
Biologics: These drugs are prescribed as infusions or self-administered injections. TNF inhibitors, such as etanercept, adalimumab, golimumab and certolizumab have been widely studied in PsA. Evidence from studies has shown that these drugs are able to control joint inflammation and skin disease, and halt radiographic progression.2
Despite their benefits, there are side effects and safety issues which can limit their use. Patients may struggle to use them as injections because of hand pain or deformity. Because of the immunosuppressive nature of TNF inhibition, patients must be screened for tuberculosis. There is a risk, albeit very low, of patients developing side effects such as congestive cardiac failure, demyelinating diseases and non-melanotic skin cancer. Hence, patients must be regularly monitored and appropriately counselled about the risks.
Some patients also experience a reduction in treatment efficacy over time and need to be switched to another agent.
Newer agents: Alongside traditional TNF inhibitors, newer biologics that target other pro-inflammatory cytokines have been developed, which may treat all facets of the disease while having a more acceptable safety profile and require less intensive monitoring. Newer agents that target different cytokines in the inflammatory pathway, such as IL-17 and IL-23, and include the interleukin inhibitors secukinumab and ustekinumab, have been developed and extensively studied in PsA patients.
In patients with predominant arthritis symptoms, TNF or IL-17 inhibitors should be considered initially when escalating treatment from csDMARDs. In patients with significant psoriasis in the context of PsA, TNF, IL-17, IL-23 or IL-12/23 inhibitors should be considered initially for treatment escalation.
Patients with predominant axial disease should be offered TNF, IL-17 or Janus kinase inhibitors as drugs with other modes of action do not have proven efficacy in axial disease.
The commencement of certain drugs depending on disease domain is largely a new approach to treatment in PsA patients.
Alongside biologics, small molecule inhibitors, such as the PDE4 inhibitor apremilast, and Janus kinase inhibitors such as tofacitinib and upadicitinib are also used. These medications are now more widely available and offer further therapeutic options for specialists if patients do not tolerate other therapies. Recent guidance from the BSR outlines how PsA patients should be managed on a step-by-step basis, although the vast majority of this guidance pertains primarily to secondary care.15
Disease monitoring
In PsA there are no universally accepted clinical scoring systems to assess disease activity, although these are under development. Patients with PsA who attend for review should have a global assessment of disease activity which includes examining both skin and joints, as well as looking for dactylitis, enthesitis and nail dystrophy. Patients should be asked to rate their pain score from 0 (no pain) to 10 (severe). The number of disease flares patients have had in between appointments should also be recorded to ensure tight disease control and treatment to target.
Routine blood tests, including full blood count, renal function, liver function tests and CRP, should be undertaken at least every three months if a patient is on disease modifying treatment, and patients should be monitored for any side effects.
Specific PsA disease activity scores have been developed in clinical trials.2 These include the PsA Disease Activity Score (PASDAS) and the Composite Disease Activity Index (CPDAI). These measures are still being evaluated in trials and are not yet used in routine clinical practice. The Minimal Disease Activity (MDA) score is another scoring system that helps assess disease activity in PsA patients. MDA is scored across seven criteria, of which five need to be fulfilled for disease to be considered minimally active (see table 2).
There is evidence to suggest that patients who present with polyarticular involvement, raised inflammatory markers and radiological joint damage at baseline are more at risk of rapid progression.2 Recent data suggest that severity of skin psoriasis is also associated with higher prevalence of joint disease.3 These factors should be taken into consideration when following up patients in primary care.
Comorbidities
PsA is associated with serious comorbidities that all clinicians should be aware of. There is an increased prevalence of cardiovascular comorbidities such as myocardial infarction and stroke in PsA patients,12 thus they should be screened for risk factors and appropriate preventive treatment initiated as appropriate.
Annual checks of height, weight, blood pressure and lipids are recommended in PsA patients. Studies have also suggested an increased risk of diabetes mellitus in PsA, partially contributed by obesity and lifestyle factors, therefore HbA1c monitoring is also of importance.13 Where appropriate, it is important to counsel patients on smoking cessation, minimising alcohol intake (this is important for patients who will usually be prescribed methotrexate as the first DMARD) and promoting weight loss at review.
PsA can also affect other organs such as the eyes; risk of uveitis is increased in PsA patients therefore clinicians should be aware of this complication and seek specialist ophthalmology input if patients present with eye redness, changes in vision or eye pain.
Conclusions
PsA is a progressive, debilitating condition which can result in considerable disability and reduced quality of life. Patients with PsA can present in a variety of ways, thus making diagnosis challenging. Early diagnosis is crucial to initiate therapy and minimise progressive joint damage.
It is a complex, challenging disease to diagnose and manage and there is evidence that it is both underdiagnosed and undertreated.16 Patients with suspected inflammatory joint disease should be thoroughly examined for any signs of psoriasis and patients with psoriasis should be regularly assessed for inflammatory joint disease.
REFERENCES
1 Ritchlin CT, Colbert RA, Gladman DD. Psoriatic arthritis. N Engl J Med 2017;376(21):2095-96
2 Haddad A, Zisman D. Comorbidities in patients with psoriatic arthritis. Rambam Maimonides Med J 2017;8(1):e0004
3 Ogdie A, Gelfand JM. Clinical risk factors for the development of psoriatic arthritis among patients with psoriasis: a review of available evidence. Curr Rheumatol Rep 2015;17(10):64
4 Wilson FC, Icen M, Crowson CS et al. Incidence and clinical predictors of psoriatic arthritis in patients with psoriasis: a population-based study. Arthritis Rheum 2009;61(2):233-39
5 Coates LC, Conaghan PG, Emery P et al. Sensitivity and specificity of the classification of psoriatic arthritis criteria in early psoriatic arthritis. Arthritis Rheum 2012;64(10): 3150-55
6 Weisman MH. Inflammatory back pain. Rheumatic Dis Clin North Am 2012;38(3):501-12
7 Gladman DD. Recent advances in understanding and managing psoriatic arthritis. F1000 Res 2016;5:2670
8 van de Kerkhof PC, Reich K, Kavanaugh A et al. Physician perspectives in the management of psoriasis and psoriatic arthritis: results from the population-based multinational assessment of psoriasis and psoriatic arthritis survey. J Eur Acad Dermatol Venereol 2015;29(10):2002-10
9 Marchesoni A, Lubrano E, Cauli A et al. Psoriatic dsease: update on traditional disease-modifying antirheumatic drugs. J Rheumatol Suppl 2015;93:61-64
10 Gossec L, Smolen JS, Ramiro S et al. European League Against Rheumatism (EULAR) recommendations for the management of psoriatic arthritis with pharmacological therapies: 2015 update. Ann Rheum Dis 2016;75:499-510
11 Coates LC, Kavanaugh A, Mease PJ et al. Group for Research and Assessment of Psoriasis and Psoriatic Arthritis 2015 Treatment Recommendations for Psoriatic Arthritis. Arthritis Rheum 2016;68:1060-71
12 Ogdie A, Yu Y, Haynes K et al. Risk of major cardiovascular events in patients with psoriatic arthritis, psoriasis and rheumatoid arthritis: a population-based cohort study. Ann Rheum Dis 2015;74(2):326-32
13 Dubreuil M, Rho YH, Man A et al. Diabetes incidence in psoriatic arthritis, psoriasis and rheumatoid arthritis: a UK population-based cohort study. Rheumatology 2014;53(2):346-52
14 Theander E, Husmark T, Alenius GM et al. Early psoriatic arthritis: short symptom duration, male gender and preserved physical functioning at presentation predict favourable outcome at 5-year follow-up. Results from the Swedish Early Psoriatic Arthritis Register (SwePsA). Ann Rheum Dis 2014;73(2):407-13
15 Tucker L, Allen A, Chandler D et al. The 2022 British Society for Rheumatology guideline for the treatment of psoriatic arthritis with biologic and targeted synthetic DMARDs. Rheumatology 2022;61(9):e255-e266
16 Holland R, Davis A, Green A et al. Psoriatic arthritis is associated with diagnostic delay and worse outcome at three months when compared to rheumatoid arthritis. Ann Rheum Dis 2017;76 Suppl 2:685
ARTICLE IN PDF
EXTERNAL WEBLINKS
For healthcare professionals
Group for Assessment and Research in Psoriasis and Psoriatic Arthritis (GRAPPA)
British Society for Rheumatology
For patients
The Psoriasis and Psoriatic Arthritis Alliance (PAPAA)

 = Registered users
= Registered users = Paid-up subscribers
= Paid-up subscribers