Doherty M, Rees F. Education the key to improving outcomes in gout. Practitioner May 2019;263(1826):11-15
Education the key to improving outcomes in gout
22 May 2019
AUTHORS
Professor Michael Doherty MA MD FRCP FHEA, Professor of Rheumatology, Department of Academic Rheumatology, School of Medicine, University of Nottingham, Nottingham, UK
Dr Frances Rees PhD MRCP, Consultant Rheumatologist, Nottingham NHS Treatment Centre, Nottingham, UK
Competing interests
Professor Michael Doherty was principal investigator for a non-drug, investigator-led, University of Nottingham study (Sons of Gout study) which was funded by AstraZeneca; and has received honoraria for ad hoc advisory boards on gout from Grunenthal and Mallinckrodt. Dr Frances Rees has no competing interests.
Article
Abstract
Gout is the most common inflammatory arthritis worldwide. In the UK, the prevalence has risen from 1.5% in 1997 to 2.5% in 2012. Gout is four times more common in men than women and prevalence increases with age. Apart from extremely painful gout flares, gout can cause joint damage with resulting chronic usage related pain and disfiguring subcutaneous tophi. Gout is also associated with comorbidities such as nephrolithiasis, chronic kidney disease (CKD), metabolic syndrome and heart disease, reduced quality of life and increased mortality. Gout is caused by precipitation of monosodium urate crystals in and around joints when the serum urate persistently exceeds saturation (> 360 µmol/L or 6 mg/dl). Urate is the end product of purine metabolism, and elevated serum urate results from increased production or reduced excretion. Increased production mainly occurs as a result of overweight or obesity but can also result from excessive intake of purine-rich foods (e.g. red meat, seafood), fructose (present in fruit juices and many fizzy drinks) or beer or spirits. Urate is mainly excreted through the kidneys and the very common heritable component of gout results predominantly from inefficient urate excretion. CKD, metabolic syndrome and drugs that affect renal function (e.g. thiazide diuretics, beta-blockers, ACE inhibitors) all reduce urate elimination. The most common presentation is a gout flare, characterised by acute onset of one, or occasionally more, hot, red, swollen joints which are very painful and tender. Less common is insidious onset of chronic usage related joint pain, especially in joints already affected by osteoarthritis. Subcutaneous tophi can also be the presentation, particulary in older people. In typical cases, clinical assessment alone is sufficient for diagnosis.
Gout is the most common inflammatory arthritis worldwide.1 in the UK, the prevalence has risen from 1.5% in 1997 to 2.5% in 2012.2 This rise is mainly attributed to increased longevity and the increased incidence/prevalence of obesity and metabolic syndrome. The incidence of gout is four times more common in men than women and its prevalence increases with age. In the UK, 15% of men and 6% of women aged 80-84 years are affected.2
As well as extremely painful flares (attacks), gout can cause joint damage with resulting chronic usage related pain and disfiguring subcutaneous nodules (tophi).
Gout is also associated with comorbidities such as nephrolithiasis, chronic kidney disease (CKD),3 metabolic syndrome, and heart disease3 as well as reduced quality of life4 and increased mortality.5
Fortunately, gout is curable. The urate crystals that cause gout can be eliminated by a combination of effective patient education and targeted urate lowering therapy (ULT).6,7 However, current management of gout in the UK is suboptimal.1,8 Although 40% of patients at diagnosis appear eligible for ULT, and 85% become eligible within five years, less than half ever receive this treatment.
Furthermore, when ULT is prescribed it is usually at a fixed dose without titration to a target serum urate level, and adherence is very poor.9 ULT prescribing ranges from 0 to 100% across practices, suggesting a wide variation in knowledge and evidence-based decision making.9
Such suboptimal management largely reflects misconceptions among both patients and doctors. Common misconceptions are that gout is not a serious condition, it is different from arthritis, and that it is self-inflicted through poor lifestyle (overindulgence in food and alcohol). Women often perceive it as a man’s disease.10,11 Hence education is key in the management of gout.
Causes
Gout is caused by precipitation of monosodium urate crystals in and around joints when the serum urate persistently exceeds saturation (> 360 µmol/L or 6 mg/dl).12
Urate crystals preferentially form in peripheral, cooler joints, especially those with osteoarthritis (changes in osteoarthritic cartilage facilitate crystal formation). Crystal deposition builds up slowly over the years without causing symptoms. However, once plentiful, some crystals on the articular cartilage surface may spill into the joint cavity (crystal shedding) and trigger intense synovitis - a gout flare. Continuing slow build-up of crystal deposits (microtophi) in cartilage and subchondral bone may also damage joints and cause symptoms and signs resembling osteoarthritis.
Gout is a common complex disorder resulting from variable interaction between genetic, constitutional and environmental factors. Urate is the end product of purine metabolism, and elevated serum urate results from increased production or reduced excretion. Increased production from endogenous purines (the main source of urate) mainly occurs as a result of overweight or obesity.
Serum urate can also rise due to excessive intake of purine-rich foods (e.g. red meat, seafood), fructose (present in fruit juices and many fizzy drinks) or beer or spirits. However, obesity is a far more important risk factor for gout than individual dietary elements. Urate is mainly excreted through the kidneys and the very common heritable component of gout is now known to result predominantly from inefficient urate excretion.
In addition, CKD, metabolic syndrome and drugs that affect renal function (e.g. thiazide diuretics, beta-blockers, ACE inhibitors) all reduce urate elimination. Heritability of gout is around 60% and recent studies show that individual dietary factors account for < 1% of serum urate variance compared with 24% from the known genetic variations already identified.13
Presentation
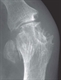
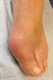
Although radiographic changes are usually a late feature, occasionally they may be seen at presentation, see figure 1. The most common presentation is a gout flare, characterised by acute onset of one, or occasionally more, hot, red, swollen joints which are extremely painful and exquisitely tender. Flares reach a peak in just 12-24 hours. Although any joint can be affected, most flares affect joints in the feet, knees, hands or elbows. Involvement of the first metatarso-phalangeal joint, termed podagra (seizing the foot), is very characteristic, see figure 2.
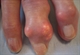
A less common presentation is insidious onset of chronic usage related joint pain, especially in joints already affected by osteoarthritis (e.g. finger interphalangeal joints with nodal osteoarthritis). Subcutaneous tophi can also be the presentation, especially in older people, see figure 3.
Tophi particularly target extensor surfaces of toes, fingers, heels, knees and elbows, appearing as nodules with heterogeneous consistency, sometimes with white-yellow discoloration (urate deposits are white).
Patients may report previous gout flares but some present solely with tophi or chronic joint symptoms. Because of the common misconceptions, mentioned earlier, patients may delay seeking advice until gout is well established.
Diagnosis
In typical cases, clinical assessment alone is sufficient for diagnosis. An acute peripheral monoarthritis that reaches its peak within 24 hours, causing what patients often describe as the ‘worst pain ever’, and which subsequently resolves completely within 1-2 weeks is virtually pathognomonic of crystal synovitis. Risk factors for gout may be apparent, such as obesity, hypertension, renal impairment, diuretic or antihypertensive drug treatment, or a family history of gout.
During a gout flare clinical examination will reveal an exquisitely tender, hot swollen joint which may be red, often with adjacent periarticular soft tissue swelling. In chronic disease asymmetrical polyarticular arthritis may be apparent, with or without tophi.
A raised serum urate helps confirm the diagnosis, though serum urate may be normal during a flare (serum urate falls during inflammation). If the joint can be aspirated, synovial fluid microscopy can demonstrate urate crystals with 100% confidence.
Radiographs do not often show characteristic features of gout until late in the disease, but joint ultrasound usually demonstrates characteristic features (microtophi in synovium and periarticular sites, and crystal deposits in superficial articular cartilage producing a parallel double contour line with subchondral bone) at first presentation in multiple peripheral sites, not just the presenting symptomatic joint.
The main differential diagnosis is acute calcium pyrophosphate crystal arthritis which is also associated with osteoarthritis but is less common. It predominates in people over the age of 60, and mainly targets the knee. Septic arthritis is a less common condition that often presents as a monoarthritis (most often the knee), but its onset is slower with symptoms and signs getting progressively worse over several days rather than just hours.
Diagnosis of gout should lead to consideration of, and screening for, associated comorbidity e.g. hypertension, hyperlipidaemia, diabetes and CKD.
Management
NICE has not yet developed a clinical guideline for gout but has endorsed the British Society for Rheumatology (BSR) recommendations that were updated in 2016.14 The following advice reflects those recommendations.
Gout flares
Flares are one of the most painful experiences known and deserve rapid treatment. All patients should be advised to rest the joint and apply ice packs.14,15 Colchicine or NSAIDS are recommended as first-line oral treatments, and should be started as soon as possible after the first symptoms appear.14
Colchicine is not an analgesic, but if started within the first 12-24 hours of the flare it can reduce crystal-induced triggering of the innate immune (inflammasome) response. It can be started at 0.5 mg twice daily and increased, if tolerated, to three times daily and then to a maximum of four times daily. Such dosing minimises the risk of side effects, especially diarrhoea which is common at higher doses.16
An alternative regimen is a 1 mg loading dose followed one hour later by 0.5 mg for the first day, and subsequently 0.5 mg twice to four times daily depending on tolerability. In patients with renal impairment the dose should be reduced to 0.5 mg daily.
Colchicine interacts with cytochrome P450 3A4 inhibitors (e.g. ciclosporin, ketoconazole, ritonavir, clarithromycin, erythromycin, verapamil, diltiazem)14 so should be used with caution in patients taking these drugs. Statins should be stopped temporarily while on colchicine.14
NSAIDs (including selective COX-2 inhibitors) should be given at maximal doses together with a proton pump inhibitor (PPI) but should be used with caution in those with renal impairment or cardiovascular disease14 and avoided in those with an eGFR < 30.
A short course of oral corticosteroid, for example prednisolone 35 mg daily,18 can be used in people with contraindications or intolerance to NSAIDs or colchicine and is often the best option for older people with multiple comorbidities.14
Joint aspiration and injection of a long-acting corticosteroid (e.g. methylprednisolone acetate or triamcinolone acetonide) gives very rapid relief and usually aborts the flare,14,19 but this option is often impractical in primary care.
Long-term treatment to eliminate urate crystals
Long-term treatment aims to cure gout by reducing and maintaining serum urate well below saturation (< 360 µmol/L). This prevents further crystal formation and gradually dissolves existing crystals, after which no further flares occur and further risk of crystal-induced joint damage is removed.
The lower the serum urate the faster the crystals dissolve and the sooner the patient is cured. The BSR recommends an initial target serum urate of < 300 µmol/L14 for the first few years of treatment. There is also growing evidence that reducing serum urate may benefit associated comorbidities, including hypertension, heart disease, CKD, that link pathogenically via elevated serum urate.14 Therefore, ULT may have additional health benefits in people with gout as well as eliminating urate crystals.
Providing individualised patient information about gout (its risk factors, associations, possible outcomes and available treatment strategies), exploring the patient’s perceptions about the illness and involving the patient in shared decision making are all key to achieving this target and maintaining adherence to ULT.6,14,20 The optimal time for patient education and engagement is once any flare has settled and the patient can fully focus on learning about their condition and its treatment. Online information and patient leaflets can be used to support this process, see External websites.
Advice on lifestyle modification should be tailored to individual needs. Where appropriate, patients should be encouraged to lose weight if overweight; reduce excessive intake of high-purine foods (e.g. red meat, offal and seafood),21 beer, spirits and fructose-rich drinks.22 Any retinol supplements should be stopped23 and consider increased intake of cherry and dairy products.21,24
For treatment of hypertension losartan and calcium channel blockers, which are uricosuric and lower serum urate, can be used in preference to beta-blockers, diuretics, ACE inhibitors and non-losartan angiotensin II receptor blockers, which elevate serum urate by reducing renal excretion.25
However, even when there are modifiable risk factors that are addressed successfully, this usually does not lower serum urate to the target level.26 Therefore to achieve cure almost all patients require ULT.14,26
Although traditionally reserved for patients with severe gout (e.g. those having several flares a year, tophi, joint damage, CKD, nephrolithiasis), recent BSR and European gout guidelines agree that ULT should be discussed with the patient at diagnosis,14,27
and even initiated following first presentation to reduce the long-term complications associated with gout. This aligns with more definitive treatment approaches used for other chronic inflammatory disease, and reflects the fact that gout is increasingly regarded as a serious health condition. When fully informed, almost all patients choose to receive ULT and subsequent adherence is excellent, see figure 4.6,20
The recommended first-line ULT is allopurinol (50-900 mg/day), a xanthine oxidase inhibitor that reduces urate production.14 The usual starting dose is 100 mg, which is then uptitrated against the serum urate level by100 mg increments approximately every 4-6 weeks until the target serum urate of < 300 µmol/L (< 360 µmol in most other countries) is achieved.
Such slow uptitration reduces the risk of severe upset, as well as possible provocation of flares associated with a rapid fall in serum urate (which partially dissolves crystals and encourages crystal shedding). Recent UK studies report 400-500 mg/day as the average dose needed to achieve target.6,20
This is higher than the 100-300 mg/day range usually prescribed in general practice.
The active metabolite of allopurinol (oxypurinol) is excreted via the kidneys so lower doses (initially 50 mg/day with subsequent 50 mg increments) should be used in patients with marked renal impairment.
Because of competitive protein binding with warfarin, slow uptitration with regular INR checks is required for patients on warfarin. Occasionally the warfarin dose will need to be reduced as ULT is uptitrated.
Most patients tolerate allopurinol, but approximately one in ten develop troublesome GI symptoms or a rash that necessitates cessation of treatment. Very rarely a serious reaction, presenting as DRESS (drug reaction or rash with eosinophilia and systemic symptoms) may develop, but this usually only occurs in patients with renal impairment who have been started on too high a dose (e.g. 300 mg), so is avoidable.
Adherence with ULT may be compromised if patients are not aware of the risk of flares continuing until all the crystals have gone. Prophylaxis to reduce flare frequency in the first months of ULT initiation may be considered, using low-dose colchicine (0.5 mg twice daily) or a daily low dose oral NSAID (plus PPI).
Whether slow uptitration of allopurinol causes a significant risk of additional flares is controversial, and when fully informed and involved in decision making, virtually all patients prefer to manage flares as required rather than take additional regular medication.6,20
Febuxostat (80-120 mg daily) is a more recently available xanthine oxidase inhibitor, which is recommended if patients are unable to tolerate allopurinol. It mainly undergoes hepatic metabolism, so no dose reduction is required with renal impairment. It has similar tolerability and side effects to allopurinol, including rarely DRESS syndrome, but there are additional concerns over its cardiovascular safety.14,27 It is available in two effective doses (80 mg and 120 mg daily) and even the lower dose may provoke flares, so there is a stronger case for considering prophylaxis.
Other alternatives are the uricosurics benzbromarone (50-200 mg daily), probenecid (250-500 mg bd) and sulfinpyrazone (200-800 mg daily), which again should be started at the lowest dose and uptitrated against the serum urate level. These drugs have limited availability, are contraindicated in significant renal impairment, and are mainly prescribed in secondary care.
Monitoring and follow-up
After ULT initiation patients should be monitored every 4-6 weeks with regular serum urate measurement until the target serum urate is achieved. Check the serum urate level again at three months, and subsequently monitor yearly to ensure serum urate is maintained < 300 µmol/L for the first 2-3 years (to speed up crystal dissolution) and long-term < 360 µmol/L (to prevent new crystal formation).
Patients may continue to experience flares during the first two or more years of successful ULT, but provided the target serum urate is maintained, gout flares will cease, any subcutaneous tophi will resolve, and quality of life is improved.
Gout cure
A recent community-based randomised trial (n = 517) compared nurse led care, involving full patient education and engagement and a treat to target ULT strategy (i.e. recommended best practice),14,27 to usual GP led care over a two-year period.20
At two years nurse led care resulted in high uptake (96%) and excellent adherence to ULT; achievement of serum urate < 300 µmol/L in 84% (< 360 µmol/L in 95%); significant reductions in flare frequency and tophi; and improved quality of life. This was achieved despite many patients having comorbidities (e.g. overweight/obesity, cardiovascular disease) and more than 20% having chronic renal insufficiency (CKD stage 3). Compared with usual care this model was cost effective at two years and estimated to be cost saving at five years.
These results highlight the importance and success of individualised education and care and treat to target ULT, principles that should be applied by any healthcare professional managing patients with gout.20
Conclusion
Gout is the only common chronic arthritis that can be cured. In primary care this can be achieved in more than 9 out of 10 patients through effective patient education and engagement and a treat to target ULT strategy.
REFERENCES
1 Kuo CF, Grainge MJ, Mallen C et al. Rising burden of gout in the UK but continuing suboptimal management: a nationwide population study. Ann Rheum Dis 2015;74:661-67
2 Kuo CF, Grainge MJ, Zhang W et al. Global epidemiology of gout: prevalence, incidence and risk factors. Nat Rev Rheumatol 2015;11:649-62
3 Kuo CF, Grainge MJ, Mallen C et al. Comorbidities in patients with gout prior to and following diagnosis: case-control study. Ann Rheum Dis 2016;75:210-17
4 Chandratre P, Roddy E, Clarson L et al. Health-related quality of life in gout: a systematic review. Rheumatology 2013;52(11):2031-40
5 Kuo CF, Grainge MJ, Mallen C et al. Effect of allopurinol on all-cause mortality in adults with incident gout: propensity score-matched landmark analysis. Rheumatology 2015;54(12):2145-50
6 Rees F, Jenkins W, Doherty M. Patients with gout adhere to curative treatment if informed appropriately: proof-of-concept observational study. Ann Rheum Dis 2013;72(6):826-30
7 Rees F, Hui M, Doherty M. Optimizing current treatment of gout. Nat Rev Rheumatol 2014;10(5):271-83
8 Cottrell E, Crabtree V, Edwards JJ et al. Improvement in the management of gout is vital and overdue: an audit from a UK primary care medical practice. BMC Fam Pract 2013;14:170
9 Kuo CF, Grainge M, Mallen C et al. Eligibility for and prescription of urate-lowering treatment in incident gout patients in England. JAMA 2014;312:2684-86
10 Spencer K, Carr A, Doherty M. Patient and provider barriers to effective management of gout in general practice: a qualitative study. Ann Rheum Dis 2012;71(9):1490-95
11 Doherty M, Jansen TL, Nuki G et al. Gout: why is this curable disease so seldom cured? Ann Rheum Dis 2012;71:1765-70
12 Chhana A, Lee G, Dalbeth N. Factors influencing the crystallization of monosodium urate: a systematic literature review. BMC Musculoskelet Disord 2015;16:296
13 Major TJ, Topless RK, Dalbeth N et al. Evaluation of the diet wide contribution to serum urate levels; meta-analysis of population based cohorts. BMJ 2018;363:k3951
14 Hui M, Carr A, Cameron S et al. The British Society for Rheumatology guideline for the management of gout. Rheumatology 2017;56(7):1056-59
15 Schlesinger N, Detry MA, Holland BK et al. Local ice therapy during bouts of acute gouty arthritis. J Rheumatol 2002;29(2):331-34
16 Terkeltaub RA, Furst DE, Bennett K et al. High versus low dosing of oral colchicine for early acute gout flare: Twenty-four-hour outcome of the first multicenter, randomized, double-blind, placebo-controlled, parallel-group, dose-comparison colchicine study. Arthritis Rheum 2010;62(4):1060-68
17 National Institute for Health and Care Excellence. Clinical Knowledge Summaries. NSAIDs - prescribing issues https://cks.nice.org.uk/nsaids-prescribing-issues [Last accessed 23 April 2019]
18 Janssens HJ, Janssen M, van de Lisdonk EH et al. Use of oral prednisolone or naproxen for the treatment of gout arthritis: a double-blind, randomised equivalence trial. Lancet 2008;371:1854-60
19 Fernandez C, Noguera R, Gonzalez JA et al. Treatment of acute attacks of gout with a small dose of intraarticular triamcinolone acetonide. J Rheumatol 1999;26(10):2285-86
20 Doherty M, Jenkins W, Richardson H et al. Efficacy and cost-effectiveness of nurse-led care involving education and engagement of patients and a treat-to-target urate-lowering strategy versus usual care for gout: a randomised controlled trial. Lancet 2018;392:1403-12
21 Choi HK, Liu S, Curhan G. Intake of purine-rich foods, protein, and dairy products and relationship to serum levels of uric acid: the Third National Health and Nutrition Examination Survey. Arthritis Rheum 2005;52(1):283-89
22 Choi HK, Curhan G. Soft drinks, fructose consumption, and the risk of gout in men: prospective cohort study. BMJ 2008;336(7639):309-12
23 Choi WJ, Ford ES, Curhan G et al. Independent association of serum retinol and beta-carotene levels with hyperuricemia: A national population study. Arthritis Care Res (Hoboken) 2012;64(3):389-96
24 Zhang Y, Neogi T, Chen C et al. Cherry consumption and decreased risk of recurrent gout attacks. Arthritis Rheum 2012;64(12):4004-11
25 Choi HK, Soriano LC, Zhang Y et al. Antihypertensive drugs and risk of incident gout among patients with hypertension: population based case-control study. BMJ 2012;344:d8190
26 Holland R, McGill NW. Comprehensive dietary education in treated gout patients does not further improve serum urate. Intern Med J 2015;45:189-94
27 Richette P, Doherty M, Pascual E et al. 2016 updated EULAR evidence-based recommendations for the management of gout. Ann Rheum Dis 2017;76:29-42
ARTICLE IN PDF
EXTERNAL WEBLINKS
Versus Arthritis
Information leaflets for patients

 = Registered users
= Registered users = Paid-up subscribers
= Paid-up subscribers